FDA-Compliant Medical Device Software
Build Software as a Medical Device (SaMD) with integrated FDA regulatory planning — from predicate research through submission support. IEC 62304, 21 CFR Part 11, and ISO 13485 built in from day one.

Let’s Discuss Your Technical Roadmap
Share a few details about your ecosystem, and we’ll prepare a preliminary integration strategy for our first call.
Why SaMD Development Requires a Different Approach
Medical device software is not just software — it’s a regulated product. The FDA classifies SaMD based on the seriousness of the healthcare situation and the significance of the information provided. Getting classification wrong early in development can mean months of rework, delayed submissions, and missed market windows.
Taction approaches SaMD projects with regulatory strategy first. Before writing a single line of code, we determine FDA classification, identify the regulatory pathway, and design the development process to produce the documentation the FDA requires.
Explore how we’ve helped hospitals, clinics, and healthcare startups
Technology Stack
Every SaMD platform we build is powered by a carefully chosen technology stack designed for reliability, traceability, and FDA compliance. We combine proven cloud-native infrastructure with regulated software development tools that produce the documentation trail the FDA expects — design history files, risk analysis records, and verification/validation evidence — so your submission is ready when your product is.
Tailored Services, Personalized Results
Turn your vision into reality. Contact us for a free quote.
Rapid Development : Experience the TURBO
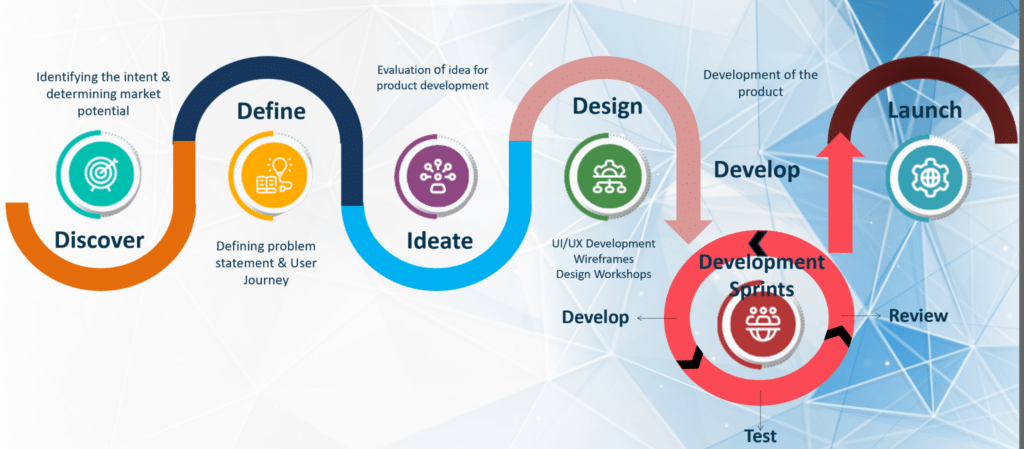
✔ Agile & User-Centric – Accelerated MVP development for faster market entry.
✔ Shortest Time-to-Market – Our TURBO framework ensures speed & efficiency.
Awards & Recognitions






Let's Collaborate. We're Just a Click Away.
Frequently Asked Questions
Still evaluating how to approach your SaMD project with FDA compliance in mind? Below are the questions medtech founders and healthcare product leaders most often ask before engaging Taction on a medical device software initiative — covering classification, regulatory pathways, timelines, and development standards. If your question isn’t answered here, schedule a free consultation and our team will walk you through your specific scenario.
